 
Calculate the molar mass of this compound. 4.A 1.78-g sample of an unknown gas has a volume of 884 mL and a pressure of 968 mmHg at 57.1 Â☌. The molar mass of NH3 (ammonia) is 17.031 g per mole. NH3 is a neutral compound therefore, the charge of NH3 is zero. Ammonia is a chemical compound that has the formula NH3, being made out of one nitrogen atom and three hydrogen atoms. Potassium nitrate, KNO3, has a molar mass of 101. 4NH3(g) + 6NO(g) â 5N2(g) + 6H2O(â) How many liters of ammonia are required to change 41.3 L of nitrogen monoxide to nitrogen gas? Assume 100% yield and that all gases are measured at the same temperature and pressure. H+-Bases: accept H+ //Ex: HCl (acid) +NH3 (base) ->Cl- + NH4+ Propane gas (C3H8) burns in. Molar mass of Ammonia (NH 3) 14.0067 g/mol + (3× 1.00794) g/mol 17.03052 g/mol 17.03052 g/mol. This value comes from the individual atoms The molar mass of KNO3 is 101. One way to remove it is to react it with ammonia. What is the rms speed of a sample of CO2 at 69.99 Â☌, in m/s? 3.Nitrogen monoxide is a pollutant commonly found in smokestack emissions. Ammonia NH3 or H3N CID 222 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety. Molecular speed can be described by the root-mean-square speed of the gas, which is the square root of the average of the squares of the speeds of all the gas molecules. 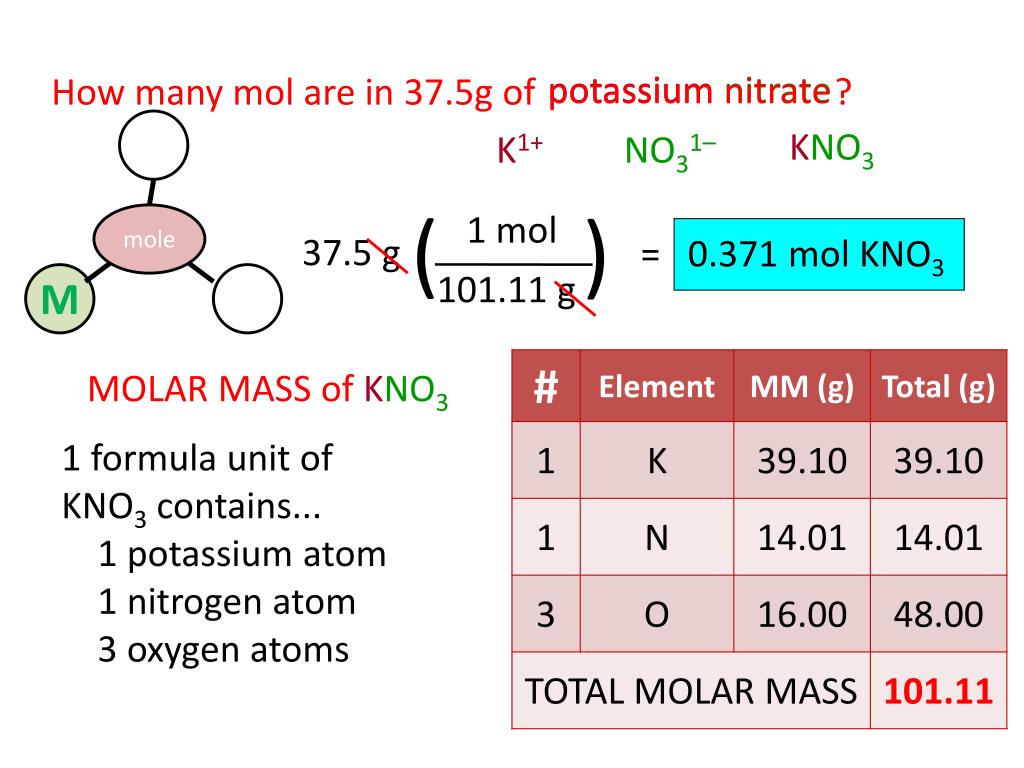
2.Molecules in a sample of a gas move at a variety of speeds. In a pair of experiments, a sample of tetrafluoroethylene, C2F4, effuses at a rate of 4.6 Ã 10-6 mol/h and an unknown gas effuses at a rate of 4.0Ã≱0-6 mol/h. Molar Mass / Molecular Weight of NH3 (Ammonia) Wayne Breslyn 635K subscribers Subscribe 560 86K views 9 years ago NH3 Lewis, Shape, Hybridization, Polarity, and more. NH3 (molar mass - 17.03 g/mol) O CHCI: (molar mass - 119.4 g/mol) SO2 (molar mass - 64.07 g/mol) O Cl (molar mass 70.90 g/mal) ON, (molar mass - 44.02 g/mol) Show transcribed image text Expert Answer Here,we can use the Ideal gas equation inorder to solve this problem. 1.Effusion is a process in which a gas escapes from a container through a small opening into a vacuum. One mole of Dinitrogen N2 and three moles of Dihydrogen H2 react to form two moles of Ammonia NH3 Show Chemical Structure Image Reaction Type Synthesis Redox Reversible reaction (equilibrium) Redox (Oxidation-Reduction) Reaction N2 + H2 NH3 might be a redox reaction.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
